

The stringent regulations from international regulatory authorities of US and EU are tightening the noose around the Indian pharma industry. The increasing inspections are driven by rising violations in the manufacturing process.
The main reason for being hauled up by a regulatory authority is the high level of risk assessment for each identified hazard. In the case of USFDA when it issues a 483s response format of inspectional observation, it is not adequately addressed by Indian companies. This leaves no option with the USFDA but to issue a warning letter. Companies do not indicate the problem instead they resort to quick fixes. Now the warning letter is perhaps a final alarm notice issued to a company before taking further legal action which includes an injunction, seizure and criminal investigation and thus drives the company out of business. The recent spurt in inspections and warning letters are adding to the woes of Indian pharma companies. Much of these issues arise because of the difficulty associated with the manufacturing process that includes not just oral formulations, sterile injectable and biologics.
There are a few characteristics that make Indian pharmaceuticals market unique:
- Branded generics dominate, making up for 70 to 80 per cent of the retail market.
- Local players have enjoyed a dominant position driven by formulation development capabilities and early investments.
- Price levels are low, driven by intense competition. While India ranks tenth globally in terms of value, it is ranked third in volumes. These characteristics present their own opportunities and challenges.
India showed the largest gains in terms of perceived ‘quality of API’ and ‘finished product’ manufacturing, proving that industry efforts to align standards more closely with the USA and Europe have clearly been noted. But perhaps most significantly, these efforts are also being transferred into reported growth potential for 2019, with India forecast to grow faster than all other major pharma economies, scoring an average of 7.16 — placing it ahead of the USA (7.04) and China (6.81). Survey respondents cited India’s ‘high-growth domestic market’ and ‘expanding manufacturing exports’ as the major drivers. The rise in exports growth potential is believed to be in response to concerted reforms by the CDSCO (Central Drugs Standard Control Organization) and industry quality improvements in the last few years.
Export trends in Indian Pharmaceuticals:
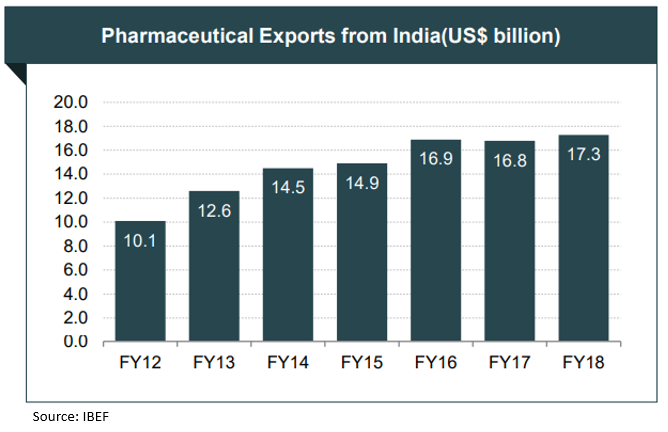
India is the world’s largest provider of generic medicines; the country’s generic drugs account for 20 per cent of global generic drug exports (in terms of volumes). Indian drugs are exported to more than 200 countries in the world, with the US as the key market. Indian pharma companies are capitalizing on export opportunities in regulated and semi-regulated markets.
Pharmaceutical exports from India reached US$ 17.27 billion in FY18. The biggest export destination for Indian pharma product is the US. In 2017, 38 per cent of India’s formulated product exports were to the US, followed by 20 per cent to sub-Saharan Africa. In FY18, 31 per cent of total pharma exports from India went to the US. Drug formulations & Biologicals is the third largest among the principal commodities exported by India during 2017-18.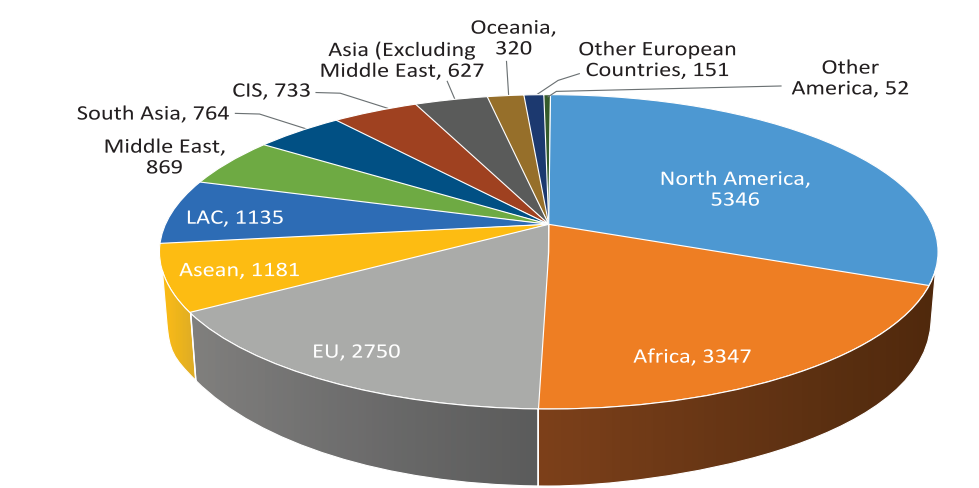
Fig: India’s Exports in 2017-18
The US is the top destination for Indian pharma exports, followed by the UK. America accounts for about 25 per cent of the country’s shipments. The other important destinations include South Africa, Russia, Nigeria, Brazil and Germany. Generic drugs form the largest segment of the Indian pharmaceutical sector, with 75 per cent market share (in terms of revenues).
India supplies 20 percent of global generic medicines in terms of volume, making the country the largest provider of generic medicines globally.
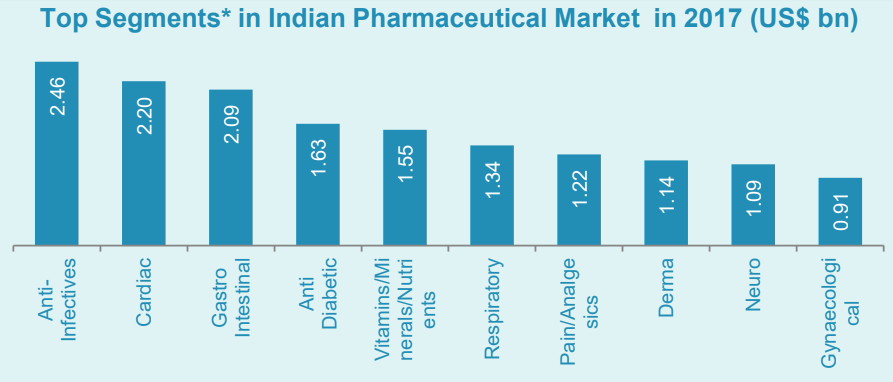
Such was the confidence in India’s pharma market, the nation was ranked joint second for ‘overall competitiveness’ of its pharma industry. India scored an average of 6.53, placing it behind only the US (6.98) and level with Germany (6.53). In addition to this, India saw the largest percentage shift in the score for quality of finished formulations, improving by 14.72 % since 2017.
Most impressively, should the same change be seen in 2019, the country’s reputation for finished product manufacturing will likely see it achieve parity with many European nations (whose scores have remained the same year-on-year) — a dramatic improvement on just a few years ago.
US-FDA Approvals by India throughout 2017-18
The USFDA approved 813 new ANDAs in 2018, the Year 2018 saw the second highest number of ANDAs granted final approval in a year, behind only the 846 approved in 2017.
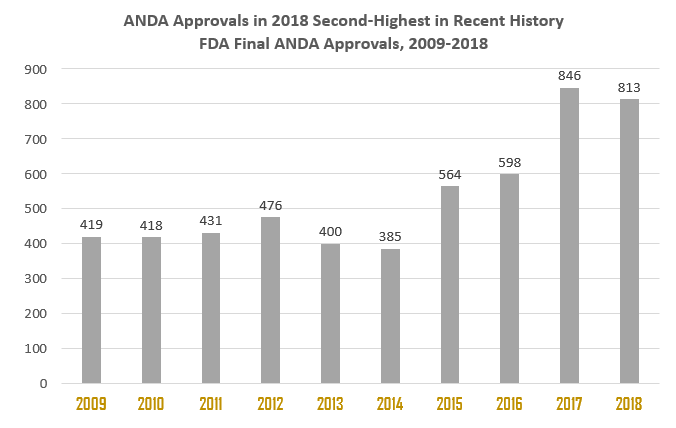
Indian Pharmaceutical companies and their subsidiaries have established a strong presence in the US during the last couple of years with higher ANDA approvals. The loss of patent exclusivity and cost-cutting measures adopted by several nations pushed the demand for the generic segment. Indian pharma companies enhanced their investments in research and development (R&D) and successfully received higher approvals from the US FDA during the last decade. Out of total 5,350 ANDA approvals between 2009 to 2018, Indian companies have secured 34.4% of these approvals and received total 1,842 ANDA final approvals. Further, out of total 1,310 tentative approvals, Indian companies grab 500 tentative approvals from USFDA which worked out to over 38.2%.
During 2018, Indian pharma companies received 290 ANDA approvals from US FDA. The USFDA approved total final 813 ANDAs in 2018, slightly lower than previous year figure of 846. Thus, Indian pharma companies captured 35.7% of total approvals in 2018. Similarly, USFDA approved total 194 tentative products during 2018 as against 174 products in the last year and Indian company’s secured 77 tentative approvals in 2018 as compared to 61 in the 2017. The overall performance was under pressure due to USFDA actions against several Indian pharma companies regarding quality and issued warning letters. However, these companies started receiving approvals after successfully resolving warning letter problem.
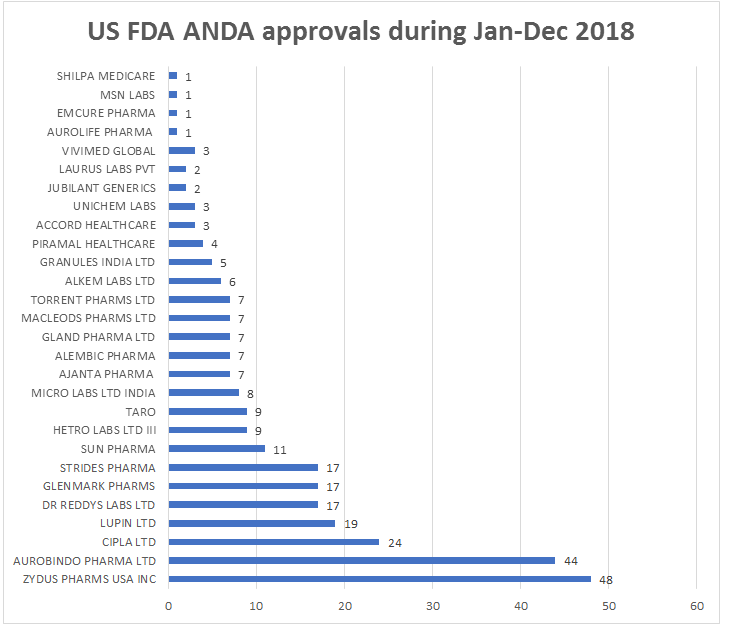
Cadila Healthcare and its US-based division Zydus Pharmaceuticals (USA) Inc. received the highest number of ANDA approvals among Indian companies during 2018. Zydus Group has received 48 final ANDA approvals followed by Aurobindo Pharma 44 and Cipla 24. Lupin got 19 ANDA approvals and Dr Reddy’s Laboratories, Glenmark Pharmaceuticals and Strides Pharma received 17 approvals each in 2018. Sun Pharma and Taro Pharmaceutical received total of 20 ANDA approvals. Among the tentative approvals, Aurobindo Pharma remained on top with 10 approvals, followed by Zydus Pharma 9 and Lupin 7. Cipla and Ajanta pharma received 6 tentative approvals during 2018. Cadila Healthcare has incurred R&D expenditure of Rs.690 crore during 2017-18 and it received 77 approval for new products for the US market.
Aurobindo Pharma, a second largest pharma company in India with consolidated net sales of Rs.16,200 crore plus, has invested Rs.667 crore during the year ended March 2018. It launched 38 products in the US, including six injectables. Cipla Rs.14,750 crore pharma giant, has continued to invest in building a strong product pipeline through R&D, which contributed significant growth in US revenues. Its annual R&D investment crosses Rs.1,000 crore and reached at Rs.1,074 crore during 2018.
Besides, these top three companies, Dr Reddy’s Laboratories, Glenmark Pharma, Sun Pharma and Strides Pharma received final ANDA approvals over 15 products each during 2018 from USFDA. Hetero Laboratories and Micro Laboratories got approvals for 9 products and 8 products each during 2018. Further, Ajanta Pharma, Alembic Pharma, Gland Pharma, Macleods Pharma and Torrent Pharma received 7 ANDA each during 2018. Alkem Laboratories and Granules India received approval for 6 and 5 products each from USFDA.
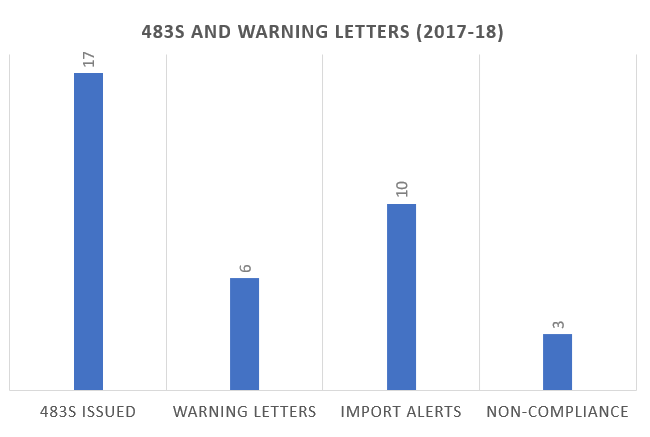
483s and Warning Letters and Import Alerts:
The number of drugs GMP warning letters continues to increase over the previous years, from 42 in FY2015 to 102 in FY2016 and to 114 in FY2017 and 127 in FY2018.
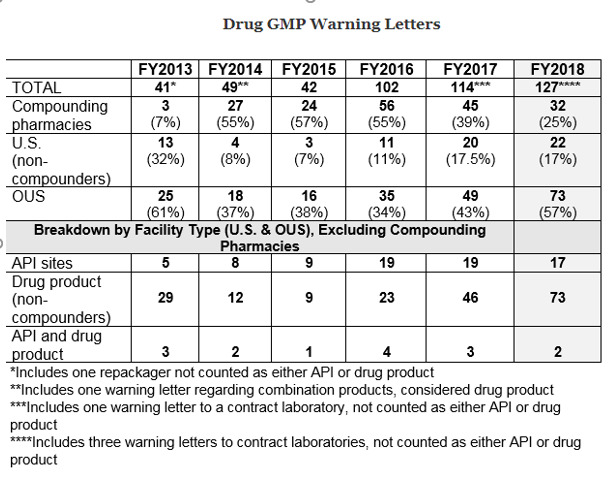
The above data shows that drug GMP warning letters have been doubled from FY2015 to FY2016 and increased again in 2017 and 2018, though not be as large a percentage.
Country wise disteibution of Drug GMP Warning Letters Issued
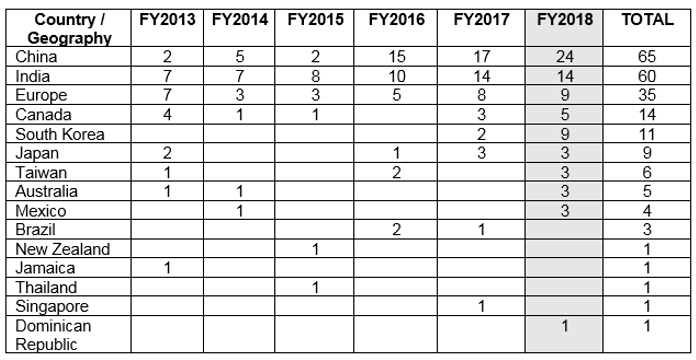
The table above shows the geographic distribution of warning letters issued outside the U.S. China received the largest number of warning letters issued to a single country in FY2018 and over the six-year period. India received the next highest number of warning letters, followed at a distance by Europe. Over the past four years, the number of warning letters to sites in China has steadily increased, while warning letters issued to sites in India have increased, then held steady.
Focus on OTC Manufacturers
Financial Year (FY) 2018 included a dramatic focus on OTC manufacturers worldwide reminiscent of the focus on compounding pharmacies and outsourcing facilities that began in 2014. Below table shows the warning letter enforcement data on this group of manufacturers for the last 11 years. The dramatic changes began in FY 2017 and increased again in FY 2018. The numbers for FY 2018 will likely increase beyond those reported here as additional warning letters issued in 2018 are posted in early 2019.
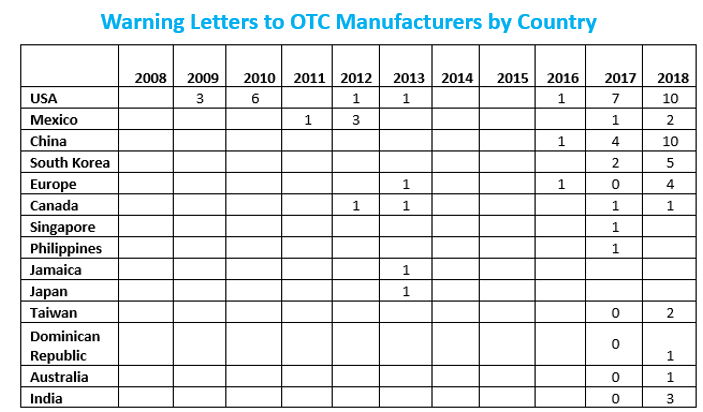
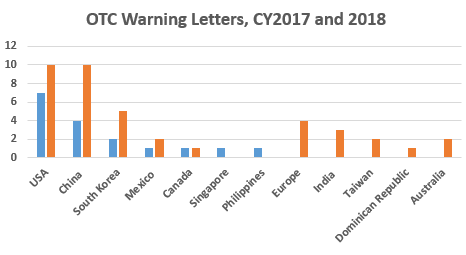
Highlighting the top deficiencies during audits by the international regulatory agencies including USFDA are:
- Inadequate quality standards
- Lack of in-house investigation on batches that fail to meet specifications
- No written processes or insufficient standard operating procedures at pharma manufacturing plants.
Not just in India but several other countries have seen these serious shortfalls. Since 2008, the number of inspections has increased but in the last six months, there has been a significant rise in the number of checks by USFDA and EMA. But the USFDA has now raised the safety concerns of many pharma manufacturers in India because it has been noted that Indian manufacturers do not give a view that their facility is on a continuous improvement process.
Further, there are clear indications of no process controls that validate the performance of manufacturing process, with nil maintenance checks on equipment cleaning, poor control of components, intermediates and raw materials besides quality assurance deficiencies. Another issue is that once the faults are pointed out by the regulator, companies submit unsatisfactory reports recording only one side of the situation with no details of the full statement that confuses the global investigators. In 2012, under the 10 drug inspection deficiencies indicated, 118 were due to poor adherence to quality processes, 78 of them were a failure to thoroughly review and 75 complaints were attributed to the absence of written procedures.
All these issues have forced the USFDA to issue warning letters because it has perceived violations to be of serious significance. The FDA does not trust the company to correct the problems sufficiently on its own and sees a lack of seriousness. Failure to establish corrective and preventive action (CAPA) following an investigation is the cause of non-conformities. Early preparation of a warning letter is likely to be based on 483s format which is a guide in the early preparedness to prevent severe lapses. The cost of an FDA enforcement action is more expensive than the cost of a robust compliance process.
Data Integrity Deficiencies in Warning Letters
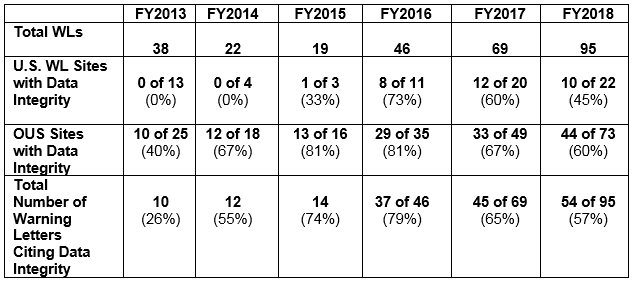
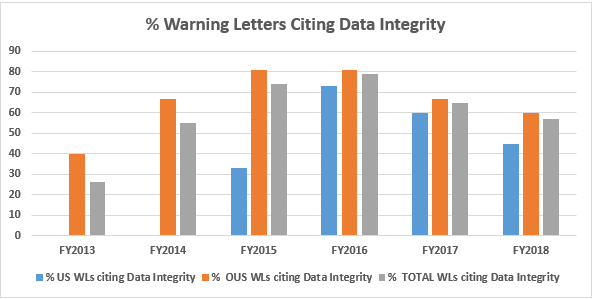
The above table shows the number of warning letters issued both inside and outside the U.S. that included references to data management and data integrity. This analysis exclude those warning letters issued to compounding pharmacies. Warning letters referencing data integrity deficiencies continue to identify the predicate rule(s) to which firms did not adhere. The percentages for both U.S. and OUS warning letters have consistently decreased between FY2016 and FY2018; perhaps we’ve turned the corner here, but we still have a long way to go to eliminate this problem.
Pharmaceuticals utilize an increasingly global manufacturing supply chain. Common approaches to organizational behavior may not always be effective due to societal considerations and there are many different influences on quality culture. We should be prepared to accept that an effective quality culture in one region may look different from that in another. By being sensitive to the influence of global culture on organizational behavior, we gain insight into the effectiveness of the ‘vigilance’ part of the quality culture, which can influence the selection of metrics. What matters is that the desired outcomes are achieved.
A strong quality culture is built upon:
- Knowledge and skills of what is important, and how a process achieves critical quality attributes
- Diligence, by fostering awareness that everyone contributes to product quality, and understanding that “my actions impact the patient and the company”
- Vigilance by individuals who know what ‘right’ and ‘wrong’ look like in their process, and a mechanism for management to be aware of problems
- Senior management commitment to being visible and transparent in decision-making so that positive outcomes can be seen from the diligence and vigilance efforts. This is more than the company mission statement – it’s ‘walking the talk’.
Impact of WTO:
The Pharmaceutical regulatory environment in India comprises of the Central Government (DCGI) and the State Government (DCO). The Drugs and Cosmetics Act, 1940 (Drugs Act) and Drugs and Cosmetic Rules, 1945 (Drug rules) regulate the import, manufacture, distribution and sale of drugs in India. Under the provisions of these Acts, the Centre appoints the Drugs Technical Advisory Board (DTAB) to advise the Central Government and the State Governments on technical matters.
In January 2005, India complied with the World Trade Organization (WTO) to follow the product patent regime known as the Trade-Related Aspects of Intellectual Property Rights (TRIPS). Before the TRIPS Agreement, most of the developed countries granted patents on drugs, but many developing countries including India only granted patents for the process of producing an invention (For example, the method of producing a drug) but not for the product (the drug itself).
As a result, generic copies of original drugs (generic drugs) were made or imported into these countries without getting permission from the patent holder. Hence, the prices of medicines were often lower because of generic competition against patented drugs. The TRIPS Agreement attempted to end this practice by implementing the International Patent Law.
From the Industrial point of view, TRIPS will give the patent holders the right to restrict their competition and therefore sell a certain drug in a monopolistic environment.
However, prices of medicines would continue to rise, making access more difficult and adversely affecting the local pharmaceutical industry. Production of generic equivalents to expensive branded drugs will be limited because of 20-year patent protection for pharmaceutical products and processes.
The new Pharmaceutical Policy, notified in 2012, intends to bring 348 essential drugs in the National List of Essential Medicines (NLEM), under the purview of the DPCO. With this policy, the market size of drugs under price control will increase from 15-20% of the domestic formulations market to 20-30%.
The government has launched two schemes—New Millennium Indian Technology Leadership Initiative in 2003 and the Drugs and Pharmaceuticals Research Programme in 1994-95—which are specially targeted at Pharmaceutical Research.
Top 10 US FDA Certified Manufacturing Plants in India with their Market Capitalization:
According to a recent study, here is a chart showing the Top 10 USFDA certified manufacturing sites in India with their Market Capitalization.
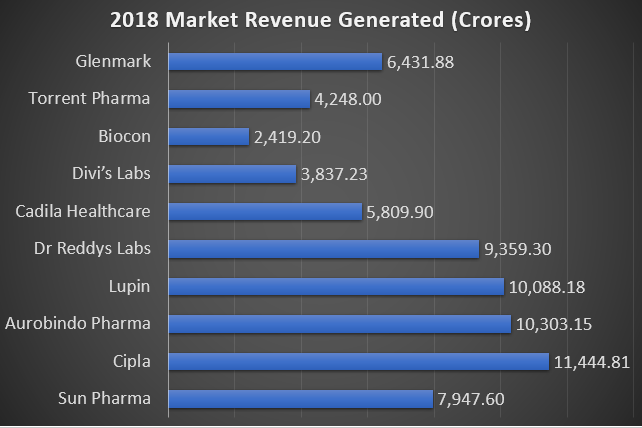
Future Opportunities and Challenges:
The Department of Pharmaceuticals has drafted ‘Pharma Vision 2020’ document, with an aim to establish India as a leading county for end-to-end drug manufacturing and innovation.
This initiative by the government aims at providing support to the Indian pharmaceutical sector through state-of-the-art infrastructure, internationally competitive scientific research personnel for pharmaceutical R&D and funding for research in the public and private sectors.
The Central government is preparing for a multi-billion-dollar investment in the pharma sector with 50% public funding through its public-private partnership (PPP) model to enhance innovation capability.
According to the ASSOCHAM, the idea is to push India into the top five pharmaceutical innovation hubs by 2020 and establish a global presence by launching one out of every 5-10 drugs discovered in India at a global level.
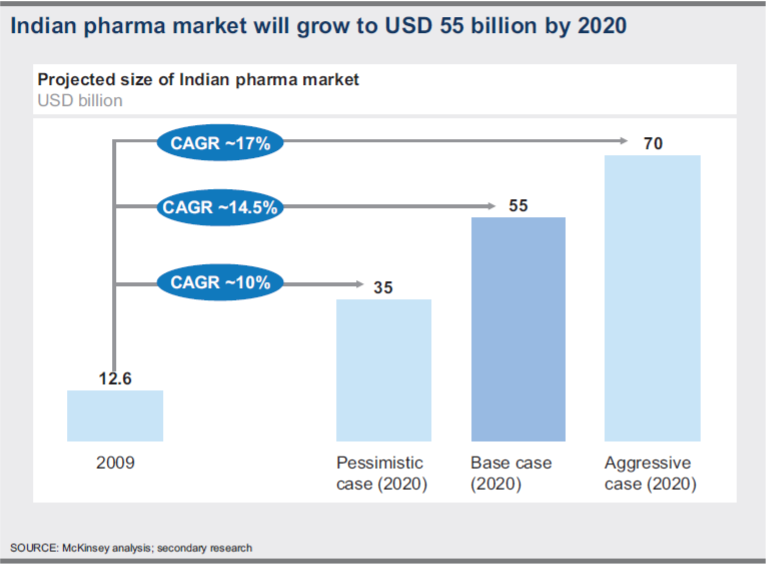
In a pessimistic scenario characterized by regulatory controls and economic slowdown, the market will be depressed and is expected to reach USD 35 billion.
In the base case scenario, all the important growth drivers will witness robust progress. The market will grow more than four times to reach USD 55 billion by 2020.
On the other hand, an aggressive case scenario, spurred by external conditions and purposeful industry actions, is definitely possible. In this scenario, GDP is expected to grow at around 8.3 per cent.
These advancements will surely put India in good competition with the regulated markets and will in turn greatly increase the export revenue.
India has made phenomenal progress in both DMF, ANDA and Biosimilar approvals over the past 7 years.
The country’s share in ANDA approvals and tentative approvals during the past years early demonstrates its aggressive ambition towards the US market. There are over 1000 molecules that provide a great opportunity for India to expand its presence in the US pharmaceutical market. India is weak in various segments such as corticosteroids, respiratory, musculoskeletal, and ophthalmic and so on and has to develop its capabilities in bio-generics for its future growth.
The pharmaceutical products from India are mostly confined to conventional tablets and capsules with very little presence in advanced formulations and completely absent from non-conventional dosage forms. India has to, therefore, urgently take corrective action to move into a higher orbit of complex chemistry, Biotech-based medicines and advanced formulations.
Overall, the exact quantum of the hit that Indian players had to deal with in the US market, varies from company to company but analysts point to many companies reporting 20 to 30 per cent decline in their US business and a price erosion to the tune of 15 to 20 per cent.
In this trend and in the overall scene there are perhaps hopes for a better 2019. Those within the industry apart from some of the leading analysts feel that things will start improving in 2019. This is linked to an expectation that many of the issues faced by Indian companies in their major export market US, will stabilize. The same also holds true for the Indian market, since in the year 2018 many companies were finally able to put behind the setbacks they had encountered on account of the Goods and Services Tax (GST) and demonetization.
Hurt by de-growth at one-point post-GST and later a single-digit growth, the domestic market growth is expected to stabilize at between 10 to 12 per cent. However, Indian pharma is also coping with cost increases in active pharmaceutical ingredients (APIs) from China, which is tightening its regulatory norms. The year also saw some significant acquisitions. For instance, IHH won the bidding war for Fortis Healthcare and Hyderabad-based Aurobindo Pharma acquired the dermatology and oral solids businesses from Sandoz Inc., USA.
As we enter the next millennium, we have to set up a vision for the New India of our dreams. This vision cannot merely be a derivative of the past. It has to be, of course, based on the reality of the present, but it has to have boldness, ambition and hope which is commensurate with the aspirations of our great nation.
We already see the winds of change. The world is going digital and also herbal. There are great opportunities for India in both areas due to its vast reservoir of intellectual capital, its rich bio-resource and its rich heritage of knowledge of the traditional medicinal system. Whereas we are already tapping into the multibillion-dollar “Digital” market, we have an opportunity to be a significant player in a multibillion-dollar “Herbal” product market too. Opportunities for becoming a global player in this market exist, but it will require an astute combination of new and traditional knowledge, innovation and creativity, and that too in the entire journey of mind to the market place.
References:
- United States Food and Drug Administration (https://www.fda.gov)
- Central Drugs Standard Control Organization.(URL: http://cdsco.nic.in/forms/Default.aspx)
- Department of Pharmaceuticals. (URL: http://pharmaceuticals.gov.in/)
- Indian Drug Manufacturers Association. (https://www.idma-assn.org/index.html)
- Indian Pharmaceutical Association. (ipapharma.org)
- Organisation of Pharmaceutical Producers of India. (https://www.indiaoppi.com/)
- Bulk Drug Manufacturers Association of India. (URL: bdmai.org)
- Directorate General of Commercial Intelligence and Statistics. (URL: http://www.dgciskol.gov.in/data_information.aspx)
- Directorate General of Foreign Trade. (URL: http://dgft.gov.in/)
- Department of Commerce. Ministry of Commerce and Industry. (URL: http://commerce.gov.in/#)
- UN Comtrade Database. (URL: https://comtrade.un.org/)
- (URL:https://www.wto.org/english/res_e/statis_e/statis_e.htm)
- International Trade Centre. (URL: http://www.intracen.org/itc/market-info-tools/trade-statistics/)
- World Integrated Trade Solution. (URL: https://wits.worldbank.org/)
- https://mhrainspectorate.blog.gov.uk/2019/02/28/quality-culture-learning-from-history/
- https://medicinman.net/2018/06/june-2018-issue/
- https://www.process-worldwide.com/report-predicts-indias-pharma-industry-to-show-strongest-growth-in-2019-a-782285/
- https://www.businesstoday.in/current/corporate/pharma-tough-year-concludes-hopes-up-for-a-better-2019/story/302136.html
- https://www.businesstoday.in/sectors/pharma/indian-pharma-is-window-of-opportunity/story/293106.html
- https://www.ibef.org/exports/pharmaceutical-exports-from-india.aspx
- https://economictimes.indiatimes.com/industry/healthcare/biotech/pharmaceuticals/pharma-exports-up-3-pc-to-17-3-billion-in-2017-18/articleshow/65338198.cms
- https://economictimes.indiatimes.com/industry/healthcare/biotech/pharmaceuticals/pharma-exports-up-3-pc-to-17-3-billion-in-2017-18/articleshow/65338198.cms
- http://pharmabiz.com/ArticleDetails.aspx?aid=111115&sid=1
- Jena et al. Presence of Indian pharmaceutical industries in US market: an empirical analysis. Journal of generic medicines. 2009; 6(4): 333-44.
- Prajapati Vishal et al. A review on drug approval process in US, Europe and India. International Journal of Drug Regulatory Affairs; 2014, 2(1): 1- 11.
- Mohidekar S et al. Common deficiencies raised by various regulatory agencies. International Journal of Drug Regulatory Affairs; 2014, 2(2): 5-6.
- US FDA approval puts India on the global map of biosimilars: Kiran Mazumdar-Shaw. (URL: http://www.forbesindia.com/article/real-issue/us-fda-approval-puts-india-on-the-global-map-of-biosimilars-kiran-mazumdarshaw/49213/1).
- Tannan S K, Badjatya J K. TRIPS and Indian pharmaceutical industry. International Journal of Drug Regulatory Affairs; 2013, 1(3): 7-13.
- Maddela L K, Insukh Oh. Advances in the review of generic drug applications by united states food and drug administration, a regulatory perspective in the era of gdufa (generic drug user fee amendment 2012). International Journal of Drug Regulatory Affairs; 2015, 3(1): 41-61.
- Express Pharma. The resurgence of pharma sector in India. (URL:http://www.expressbpd.com/pharma/cover-story/the-resurgence-of-pharma-sector-in-india/395177/)
- India: Indian Pharmaceutical Companies: Abbreviated New Drug Application Approvals2017.(URL:http://www.mondaq.com/india/x/674750/Life+Sciences+Biotechnology/Indian+Pharmaceutical+Companies+Abbreviated+New+Drug+Application+Approvals+2017)
- Mid 2018 – Recap of Warning Letters, Import Alerts and Non-compliances. (URL: https://www.pharmacompass.com/radio-compass-blog/mid-2018-recap-of-warning-letters-import-alerts-and-non-compliances)
- An Analysis Of FDA FY2016 Drug GMP Warning Letters. (URL: https://www.pharmaceuticalonline.com/doc/an-analysis-of-fda-fy-drug-gmp-warning-letters-0001)
- Top 10 Pharma companies in India. (URL: http://listz.in/top-10-pharma-companies-in-india.html)
- India Brand Equity Foundation. Indian Pharmaceutical Industry Analysis. (URL: https://www.ibef.org/industry/indian-pharmaceuticals-industry-analysis-presentation)
- Economic Times. Will India achieve PharmaVision 2020. (URL: https://economictimes.indiatimes.com/opinion/et-commentary/will-india-achieve-pharma-vision-2020/articleshow/28159936.cms)
- ASSOCHAM India. India will be one of top 5 global pharma innovation hubs by 2020 through its PPP model. (URL: http://www.assocham.org/newsdetail.php?id=5886)
- The Week. India to be among the top 5 pharma innovation hubs by 2020. (URL: https://www.theweek.in/content/archival/news/biz-tech/india-to-be-among-top-5-pharma-innovation-hubs-by-2020.html)
- IMARC Research. FDA warning letters and (URL:http://www.imarcresearch.com/blog/bid/280993/fda-warning-letters-and-form-483s-what-s-the-difference)
- Department of Pharmaceuticals. Annual report 2016-17. (URL: http://pharmaceuticals.gov.in/sites/default/files/Chemical%20Pharma%20-2016-17.pdf)
- India’s Export trend from April 2009 to March 2017. (URL: http://pharmabiz.com/services/ExportImport/India_export_2009-2017.pdf)
- India’s Leading Pharmaceutical Companies. (URL: http://www.dnb.co.in/Publications/LeadingPharmaceutical2016/IndiasLeadingPharmaceuticalCompanies2016.pdf)
- India Pharma 2020 Propelling access and acceptance, realising true potential. (URL:http://online.wsj.com/public/resources/documents/McKinseyPharma2020ExecutiveSummary.pdf)
- India Brand Equity Foundation. Pharmaceuticals report July 2018. (URL: https://www.ibef.org/download/Pharmaceuticals-March-2018.pdf)
- US drug approvals are through the roof. (URL: https://qz.com/1172576/usfda-approvals-are-through-the-roof-and-indian-pharma-companies-are-worried/)
- Indian firms get FDA approval for 110 generic drugs. (URL: https://www.thehindu.com/business/Industry/indian-firms-get-fda-approval-for-110-generic-drugs/article5130296.ece)
Pharma Knowledge Centre – “My Learning Life” (PKC) engaged Young Indian Youths to prepare this presentation/Blog and the same has been prepared by one of the associated youngsters in consultation with PKC.
All rights reserved. All copyright in this presentation and related works is solely and exclusively owned by PKC. The same may not be reproduced, wholly or in part in any material form (including photocopying or storing it in any medium by electronic means and whether or not transiently or incidentally to some other use of this presentation), modified or in any manner communicated to any third party except with the written approval of PKC.
This presentation is for information purposes only. While due care has been taken during the compilation of this presentation to ensure that the information is accurate to the best of associate and PKC’s knowledge and belief, the content is not to be construed in any manner whatsoever as a substitute for professional advice.
Associate and PKC neither recommend nor endorse any specific products or services that may have been mentioned in this presentation and nor do they assume any liability or responsibility for the outcome of decisions taken as a result of any reliance placed on this presentation.
Neither any associate nor PKC shall be liable for any direct or indirect damages that may arise due to any act or omission on the part of the user due to any reliance placed or guidance is taken from any portion of this presentation.

