

The sole purpose of a Medical Insurance is to help the insured in times of unexpected tragedies. Insurance, as a business, is a very profitable one and that is why so many industrial houses and banks have entered this sector in the last two decades.
Clinical Trial is a highly structured and regulated scientific study where the Safety and Efficacy of a new drug product is tested in Human subjects. Since the outcome of a such experiments cannot be accurately predicted, the fact that the human volunteer in the study may suffer potential health hazards is inevitable. It is because of this risk involved in conducting Clinical trials, these studies involve huge investments – on an average it takes about 1 Billion Dollars and 10 years of research to bring a new drug to market.
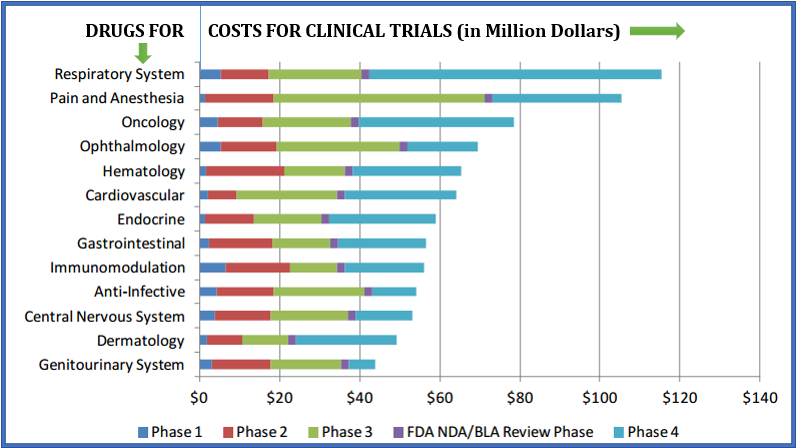
Insurance issues in Clinical trial development phases are usually not considered as a priority in research ethics. Compensation for injuries in clinical trials is required in many countries where clinical trials are conducted; however according to recent medical studies, the procedures and rules differ significantly. Clinical Research participants engage in experimentations which will hopefully provide useful results for future patients and need not to be harmed as a consequence. Thus, the availability of a compensation for every research participant at all the clinical development phases, testifies reciprocity on the part of society, to compensate the volunteers for the risks they are undertaking.
Under the Indian rules, the compensation may vary from Rs. 4 lakhs to Rs. 73.6 lakhs. Although the ethical arguments for compensating injured participants are generally acknowledged, the practical details are complicated – particularly the Determination of the Extent and Duration of coverage and the Assignment of Responsibility for Paying Compensation. This applies especially in the oncology clinical trials where phase 1 oncology trials involve healthy patients. The misinterpretation about cancer clinical trials are frequent among trial participants, and the Principal Investigator might also prevail in some of these misconceptions. Compensation may depend on the kind of trial the subjects are participating in. These can be early clinical development phases (phase 0 clinical trial, phase 1 and phase 2) or trials with already tested products (phase 3 and phase 4).
Indian Pharmaceuticals market is expected to touch US$ 55 billion by 2020 from the current level of US$ 36.7 billion in 2016 growing at a compound annual growth rate (CAGR) of 15.92 per cent, according to ASSOCHAM-IITTM joint study, but the role of sponsors in providing medical treatment of ‘Clinical Trial Related Injuries and Death’ has long been a topic of intense debate.
“The experiment should be so conducted as to avoid all unnecessary physical and mental suffering and injury”
- The Nuremberg Code
Although all the policies generally have some deficiencies, in one respect or the other, none have a provision to pay full compensation if required. While some sponsors follow the rules that exist, often the compliance is in the letter and not in the spirit. Some insurers put in some of the ‘preconditions’ that jeopardize the payment of compensation to subjects. It is finally the responsibility of sponsors to pay compensation, irrespective of whether their insurance covers it or not. The Ethical Committees must intervene at all the stages of a Clinical Trial and ensure that the subjects’ rights and well-being are protected. Patients who volunteer to participate in clinical trials deserve to know that their contribution to improving human health will be available to inform health care decisions. The knowledge made possible by their collective altruism must be accessible to everyone.
References:
- Gainotti S, Petrini C (2010) Insurance Policies for Clinical Trials in the United States and in some European Countries. J Clinic Res Bioeth 1:101 doi:10.4172/2155-9627.1000101.
- Steven Joffe, E Francis Cook, Paul D Cleary, Jeffrey W Clark, Jane C Weeks. Quality of informed consent in cancer clinical trials: a cross-sectional survey. The Lancet. 2001;358(9295): 1772-77.
- Ghooi, Ravindra B., and Deepa Divekar. “Insurance in Clinical Research.” Perspectives in Clinical Research4 (2014): 145–150. PMC. Web. 23 July 2018.
- The Associated Chambers of Commerce & Industry of India (ASSOCHAM). Accessed on: 23 July 2018.
URL:http://www.assocham.org/newsdetail.php?id=6208
- Cathrine D A, Jeffrey M, et. al. Clinical trial registration: a statement from the international committee of medical journal editors. N. Engl. J. Med. 351(12): 1250-51.
- Trials of War Criminals before the Nuremberg Military Tribunals under Control Council Law No. 10″, Vol. 2, pp. 181-182. Washington, D.C.: U.S. Government Printing Office, 1949.
Image source: U.S. Department of Health & Human Services: Office of The Assistant Secretary for Planning and Evaluation. Examination of Clinical Trial Costs and Barriers for Drug Development (URL: https://aspe.hhs.gov/report/examination-clinical-trial-costs-and-barriers-drug-development)

